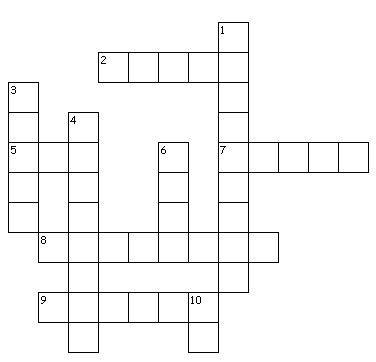
Using your knowledge of Atomic Structure, solve the following Criss Cross Puzzle. This activity is to orient you to the next topic, which is Ionic Bonding. We will start on this in the next lesson (Monday, 31st March)! Click "Read More" to attempt the puzzle!
Across
2. The maximum number of electrons in the second and third shell
5. The maximum number of electrons in the first, innermost shell
7. A nonmetal atom that has achieved a stable configuration is called an _____.
8. Identical charges are ________ by each other.
9. An atom that loses electrons to achieve a stable configuration is called a ______.
Down
1. Opposite charges are _________ to each other.
3. An atom has two shells and 1 valence electron. Is this a metal or a nonmetal atom?
4. An atom has two shells and 7 valence electrons. Is this a metal or a nonmetal atom?
6. Table salt is an ionic compound. What is its Chemical Formula?
10. An ion has two shells and 8 valence electrons. Can you identify the ion?
Pen your responses in the Comment section below!
2. The maximum number of electrons in the second and third shell
5. The maximum number of electrons in the first, innermost shell
7. A nonmetal atom that has achieved a stable configuration is called an _____.
8. Identical charges are ________ by each other.
9. An atom that loses electrons to achieve a stable configuration is called a ______.
Down
1. Opposite charges are _________ to each other.
3. An atom has two shells and 1 valence electron. Is this a metal or a nonmetal atom?
4. An atom has two shells and 7 valence electrons. Is this a metal or a nonmetal atom?
6. Table salt is an ionic compound. What is its Chemical Formula?
10. An ion has two shells and 8 valence electrons. Can you identify the ion?
Pen your responses in the Comment section below!